The Sunoco/UOP phenol process produces high-quality phenol and acetone by liquid-phase peroxidation of cumene.
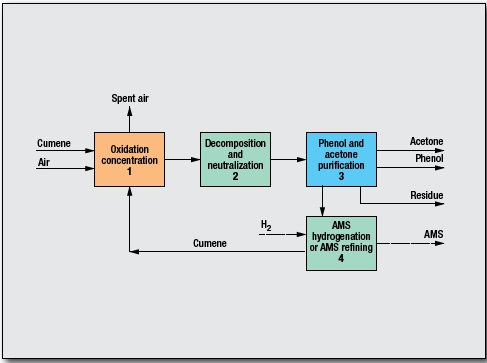
Key process steps:
Oxidation and concentration (1): Cumene is oxidized to cumene hydroperoxide (CHP). A small amount of dimethylphenylcarbinol (DMPC) is also formed, but low-pressure and low-temperature oxidation results in very high selectivity of CHP. CHP is then concentrated and unreacted cumene is recycled back to the oxidation section.
Decomposition and neutralization (2): CHP is decomposed to phenol and acetone, accompanied by dehydration of DMPC to alphamethylstyrene (AMS), catalyzed by mineral acid. This unique design achieves a very high selectivity to phenol, acetone and AMS without using recycle acetone. Decomposed catalyst is neutralized.
Phenol and acetone purification (3): Phenol and acetone are separated and purified. A small amount of byproduct is rejected as heavy residue.
AMS hydrogenation or AMS refining (4): AMS is hydrogenated back to cumene and recycled to oxidation, or AMS is refined for sale.
Cumene peroxidation is the preferred route to phenol, accounting for more than 90% of world production. The Sunoco/UOP Phenol process features low feedstock consumption (1.31 wt cumene/wt phenol). High phenol and acetone product qualities are achieved through a combination of minimizing impurity formation and efficient purification techniques. Optimized design results in low investment cost along with low utility and chemicals consumption for low variable cost of production. No acetone recycle to the decomposition section and simplified neutralization make the Sunoco/UOP Phenol process easier to operate.
Licensor: Sunoco and UOP LLC, A Honeywell Company